Examples of paramagnetic elements

Rachel Hickman
Published Jan 01, 2026
What are examples of paramagnetic materials?
Paramagnetic Materials: These are metals that are weakly attracted to magnets. They include aluminum, gold, and copper. The atoms of these substances contain electrons most of which spin in the same direction but not all . This gives the atoms some polarity.What are the paramagnetic elements?
Iron oxide, FeO, has a very high value of 720. Other materials considered strongly paramagnetic include iron ammonium alum (66), uranium (40), platinum (26), tungsten (6.8), cesium (5.1), aluminum (2.2), lithium (1.4) and magnesium (1.2), sodium (0.72) and oxygen gas (0.19).Is o2 − paramagnetic?
Oxygen is paramagnetic mainly because it consists of two unpaired electrons in its last molecular orbital. This can be proven if we look at the molecular orbital diagram of oxygen.How do you know if an element is paramagnetic or diamagnetic?
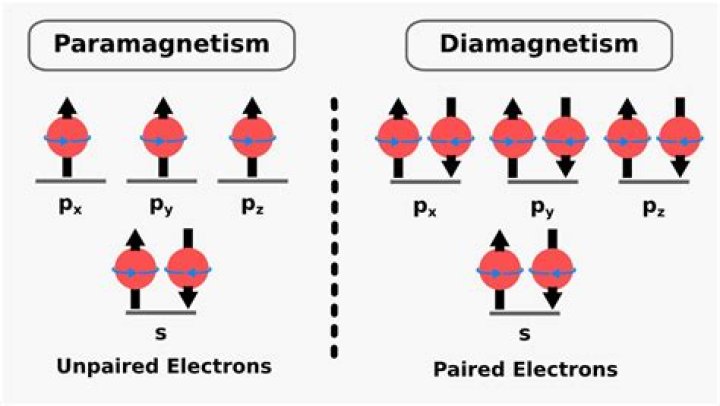
The magnetic properties of a substance can be determined by examining its electron configuration: If it has unpaired electrons, then the substance is paramagnetic and if all electrons are paired, the substance is then diamagnetic.Which of the following is a paramagnetic?
Step by step solution by experts to help you in doubt clearance & scoring excellent marks in exams. Paramagnetic species contains unpaired electrons in their molecular orbital electronic configuration. Thus, among the given species only O-2 is paramagnetic.Is Cu+ paramagnetic or diamagnetic?
Since the Cu+ ion has no unpaired electrons, hence it is diamagnetic.Is Iron diamagnetic or paramagnetic?
Compounds in which all of the electrons are paired are diamagnetic.6.3 Paramagnetic Properties of Fe, Fe2+ and Fe. 3+
| Sample | Paramagnetic? |
|---|---|
| Iron(III) | Paramagnetic (1 lone electron). |
| Salt | Diamagnetic. |
| Sugar | Diamagnetic. |
| Water | Diamagnetic. |



